MAMS
A multi-arm, multi-stage (MAMS) platform to accelerate our search for a cure.
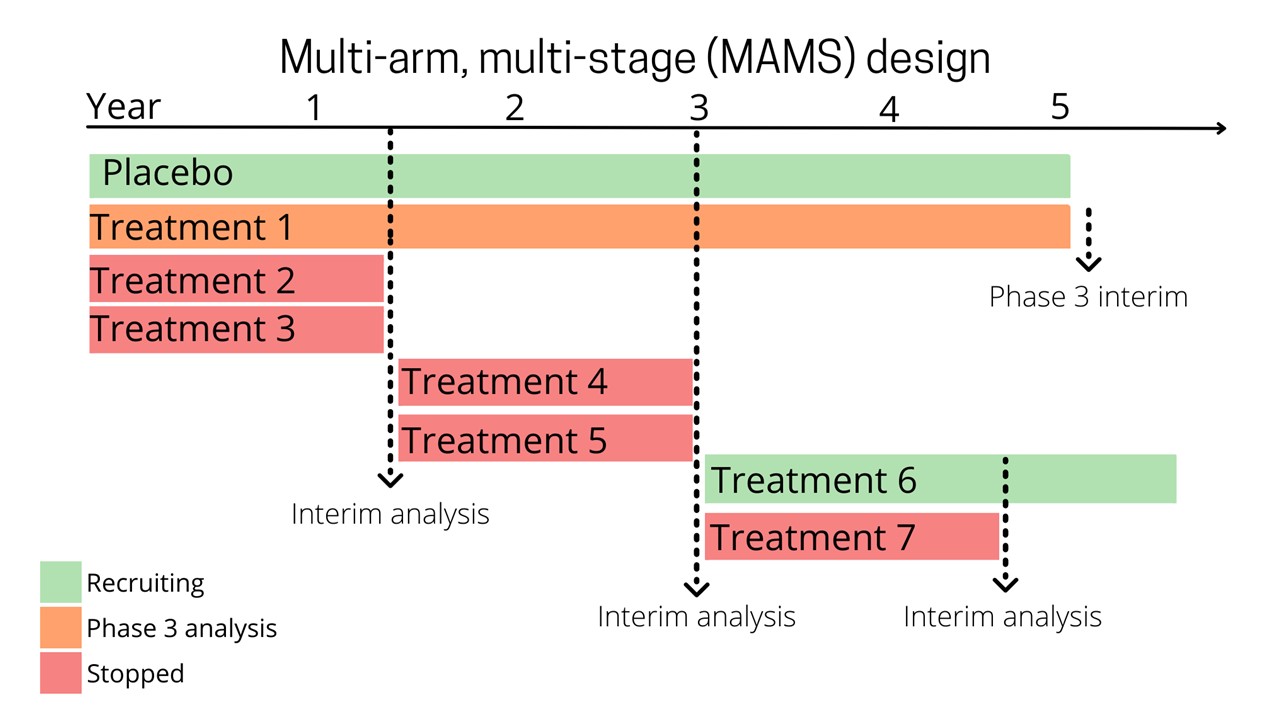
What is a MAMS platform trial?
A multi-arm, multi-stage (MAMS) platform trial is an innovative approach to clinical trials. MAMS designs tests multiple treatments at once.
In current clinical trials, potential treatments need to pass three phases of clinical testing to determine their safety and efficacy. Each time a phase ends, the trial infrastructure is dismantled and has to be rebuilt for the next phase. This takes a lot of time and resources. Currently, it can take more than 10 years for one treatment to go through this process, slowing down the evaluation of potential new treatments.
With a MAMS trial, multiple treatments are tested at once, in the same trial against one placebo.
There are regular ‘interim’ checkpoints to test whether a treatment appears to be working. If a treatment does appear to be working, then recruitment will continue until there is enough participants to determine whether the treatment is effective.
If a treatment does not appear to be effective, then recruitment to that arm will be stopped. A new potential treatment will replace it, without needing to stop the trial.
This has multiple advantages including getting answers about multiple treatments quickly, needing fewer patients to be treated with placebo, saving time and resources and overall being more cost-effectiv
